Acids and Bases (2 of 2)
Part 1 of Acids and Bases is located here.
Dissociation Constant
At equilibrium, the rate of dissociation of the product AB is equal to the rate of associate of the reactant A and B
∴ at equilibrium:
Kd = [A] [B] / [AB]
Which is derived from an example such as: AB ↔ A + B @ Equilibrium
pH Indicators
pH indicators work because of the dissociation of the H+ ions in the indicator itself.
| Common Indicators | Range |
| Phenolphthalein | 8 – 10 |
| Methyl Red | 4.5 – 6 |
| Bromothymol | 6 – 7.6 |
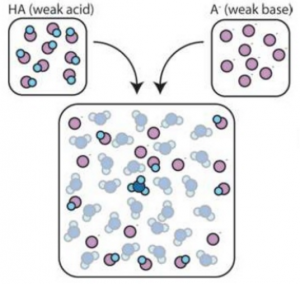
Buffer Solutions
Buffer solutions resist changes in pH.
The buffer solution absorbs the proton
Example:
HA + H2O ↔ H3O+ + A–
Ka = [H3O+] [A–] / [HA]
-log(Ka) = -log([H3O+] ([A–] / [HA]))
-log(Ka) = -log([H3O+]) – log([A–] / [HA])
pKa = pH – log([A–] / [HA])
pH = pKa + log([A–] / [HA])





You must be logged in to post a comment.